
One molecule of vitamin C is made up of 20 atoms (6 carbons, 8 hydrogens, and 6 oxygens - that's C 6H 8O 6). Look at that molecule of water again - each hydrogen has one bond, and the oxygen in the middle has two bonds. For example, a hydrogen atom forms one bond, an oxygen atom forms two, and carbon forms four bonds. Now, if you stick an oxygen atom in between those two hydrogens, you have a molecule of water - H 2O!Įach type of atom typically forms the same number of bonds (they tend to be stubborn that way). Pretty simple, huh?Ĭlick here to take a look at a 3-D model of hydrogen (H 2). For example, two atoms of hydrogen hook together to form a molecule of hydrogen, H 2 for short. If it's in the table, it's an element!Ītoms can join together - they form bonds together - to make MOLECULES. (We can call them by their names, or by their symbols - C for carbon, H for hydrogen, and O for oxygen.) All the elements are listed out in a periodic table. Carbon is an element, hydrogen is an element, and so is oxygen. Solids, liquids, gases - all matter - are made up of atoms (or other things, like molecules, that are made from atoms)!ĮLEMENTS are the kinds of atoms that we can have. Atoms are so small that there are millions and billions and trillions in the tiniest speck you can see. ATOMS are the basic building blocks of everything you can see around you, and even lots of things you can't see, like the air that you breathe. Let's talk tiny, and I mean really itty-bitty. OK, so - what's an atom and what does it have to do with molecules?  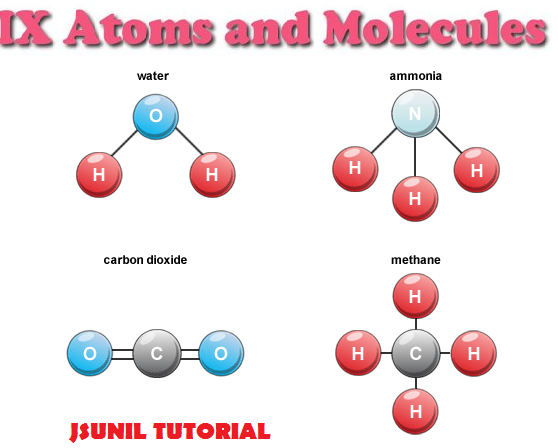
If you already know what ATOMS and ELEMENTS and MOLECULES are and want to dive straight into polymers, click here.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
